Phase Diagram And Heating Cooling Curves

Heating And Cooling Curves вђ Overview Examples Expii Boil water. heat steam from 100 °c to 120 °c. the heat needed to change the temperature of a given substance (with no change in phase) is: q = m × c × Δ t (see previous chapter on thermochemistry). the heat needed to induce a given change in phase is given by q = n × Δ h. using these equations with the appropriate values for specific. By removing the time axis from the curves and replacing it with composition, the cooling curves indicate the temperatures of the solidus and liquidus for a given composition. this allows the solidus and liquidus to be plotted to produce the phase diagram: this page titled 12.5: interpretation of cooling curves is shared under a cc by nc sa.
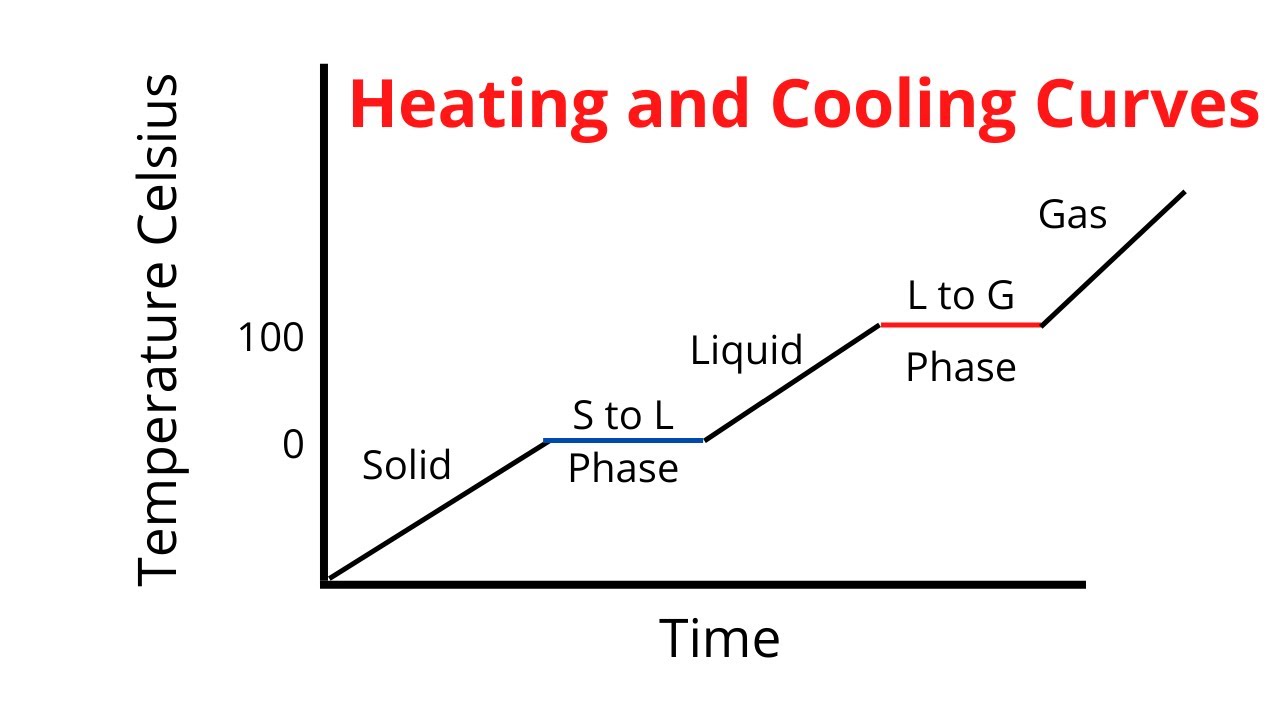
Heating And Cooling Curve Introduction Plus Kinetic And Potential Heating and cooling curves are graphs. they plot a substance's temperature (y axis) against heat (x axis). for heating curves, we start with a solid and add heat energy. for cooling curves, we start with the gas phase and remove heat energy. cooling and heating curves have five segments. The experiment described above can be summarized in a graph called a heating curve (figure below). figure 13.18.1 13.18. 1: in the heating curve of water, the temperature is shown as heat is continually added. changes of state occur during plateaus, because the temperature is constant. Also described was the use of heating and cooling curves to determine a substance’s melting (or freezing) point. making such measurements over a wide range of pressures yields data that may be presented graphically as a phase diagram. The compound cholesteryl benzoate is a rod like molecule that undergoes a phase change from the solid to the liq uid crystal phase at 145.5 °c. when cholesteryl benzoate is mixed with cholesteryl oleyl carbonate, a molecule with a curved shape, the temperature of the solid to liquid crys tal transition changes.

Heating And Cooling Curves Read Chemistry Ck 12 Foundation Also described was the use of heating and cooling curves to determine a substance’s melting (or freezing) point. making such measurements over a wide range of pressures yields data that may be presented graphically as a phase diagram. The compound cholesteryl benzoate is a rod like molecule that undergoes a phase change from the solid to the liq uid crystal phase at 145.5 °c. when cholesteryl benzoate is mixed with cholesteryl oleyl carbonate, a molecule with a curved shape, the temperature of the solid to liquid crys tal transition changes. Phase diagrams (plots of pressure vs. temperature) were correlated with heating curves (plots of temperature vs. energy). these two types of plots provide complementary information on the phase transitions of substances. while a heating curve provides information on the phase changes at a single pressure, the phase diagram depicts the phase. Understanding heating and cooling curves is crucial for grasping how substances absorb or release heat during phase changes. as a substance heats up, it undergoes an endothermic process, indicated by a positive heat variable (q), absorbing energy to break molecular bonds and transition from solid to liquid (melting or fusion) and eventually to gas (vaporization).
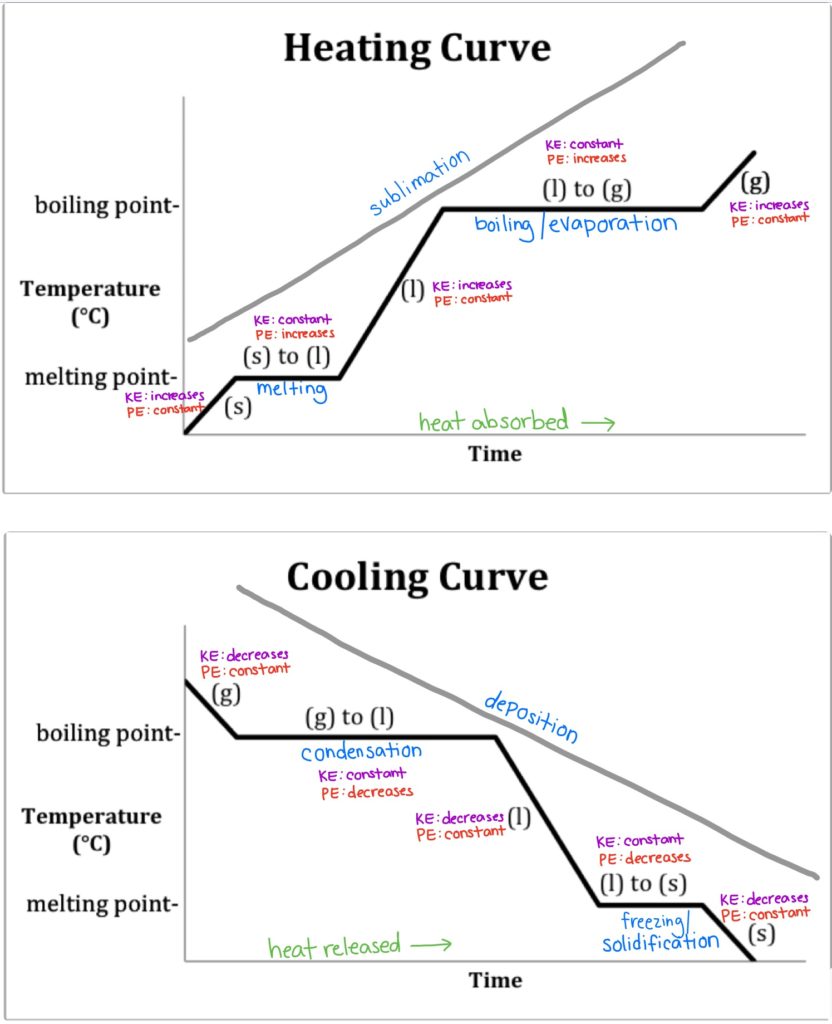
Heating And Cooling Curves Phase diagrams (plots of pressure vs. temperature) were correlated with heating curves (plots of temperature vs. energy). these two types of plots provide complementary information on the phase transitions of substances. while a heating curve provides information on the phase changes at a single pressure, the phase diagram depicts the phase. Understanding heating and cooling curves is crucial for grasping how substances absorb or release heat during phase changes. as a substance heats up, it undergoes an endothermic process, indicated by a positive heat variable (q), absorbing energy to break molecular bonds and transition from solid to liquid (melting or fusion) and eventually to gas (vaporization).

Comments are closed.